Graphite is More Than Just Pencil Lead
by Jenny Knodell, IQS Editor

When asked about the mineral graphite, one image likely comes to mind: a pencil. This soft, dark grey, greasy material, also called black lead and plumbago, has been used to jot down notes and draw pictures for hundreds of years. Its name is even derived from the Latin term for “to draw/write.” Graphite is only found in a handful of places around the world, including Africa, the British Isles and a couple of U.S. states—including my home state of Michigan. There are 3 main types of natural graphite that are all found in different types of ore deposit. Crystalline graphite is flat with hexagonal edges, amorphous graphite is very thin and flakey, and lump graphite is found in veins of fractures underground. The first pencil was composed of sticks of graphite tied together with string. As you can imagine, this early writing utensil wasn’t exactly user friendly, but it could easily be erased with rubber. It wasn’t until much later that the modern pencil—the wooden kind with a pink eraser top that we use today—became popular.

Raw natural graphite and lead pencil.
The lead we use in pencils is actually a mixture of graphite and clay or wax. The process that transforms raw lead and wood into a pencil is a little more complicated than it seems. First, graphite is ground up into find powder and added to clay or wax and water in a mixer. The water is squeezed out, and the mixture is left to dry. Water is then added a second time to the powder. A thick paste is formed, and extruded through thin metal tubes to become the thin, round lead pencil shape. The wood components of the pencil are cut as halves and glued around the lead. Each pencil has a designated hardness level—most are #2, but the scale may reach 4. This number determines how much graphite is released onto the page per pencil stroke. The higher the number, the less graphite is released and the lighter the pencil line will be. After the wood part is cut to shape and painted, the eraser and metal part are added last, along with the embossed hardness number and logo. And there you have it. A pencil is born.
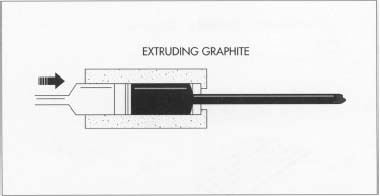
Extruding graphite for pencil lead.
Although pencil lead may be the most widely known and popular use for graphite, many other graphite products exist. Since graphite is so heat resistant, it is often used to make crucibles, containers that hold material at very high temperatures without being burned, melted or chemically changed. In steelmaking, graphite is used to lubricate the molds used in steel extrusion. It is also sometimes found in paint that coats the inside of a mold, which is used to ease the ejection of a finished molded product. Carbon graphite is often a cheaper alternative to carbon raisers, which are added to steel to raise the carbon content. Synthetic graphite, which is made by heating coal, also finds uses in some other industrial applications. Since man-made graphite is electrically conductive, graphite electrodes are used to heat electric arc furnaces. All graphite, whether synthetic or natural, is easily 100% recyclable. Trim and excess graphite from product manufacturing often finds a second life in electric arc furnaces.

Graphite Electrodes. Photo courtesy of Weaver Industries, Inc.
Graphite products. Photo courtesy of Becker Brothers Graphite Corporation.


 Castings & Forgings
Castings & Forgings Bulk Material Handling
Bulk Material Handling Electrical & Electronic Components
Electrical & Electronic Components Flow Instrumentation
Flow Instrumentation Hardware
Hardware Material Handling Equipment
Material Handling Equipment Metal Cutting Services
Metal Cutting Services Metal Forming Services
Metal Forming Services Metal Suppliers
Metal Suppliers Motion Control Products
Motion Control Products Plant & Facility Equipment
Plant & Facility Equipment Plant & Facility Supplies
Plant & Facility Supplies Plastic Molding Processes
Plastic Molding Processes Pumps & Valves
Pumps & Valves Recycling Equipment
Recycling Equipment Rubber Products & Services
Rubber Products & Services